Our lab studies how metabolism contributes to animal development, with a particular focus on lipid droplets (LDs). LDs are fat storage organelles with critical roles in both lipid and protein metabolism, but whose role in development was previously largely unexplored. Using fruit flies as our model, we find that LDs promote the maturation of oocytes, regulate the timing of embryogenesis, and control the embryonic transcriptome. They do so via multiple pathways that involve intracellular motility, metabolism, and protein sequestration. We are also studying roles for LDs in immunity and how the transcription factor HSF regulates development.
Protein Sequestration
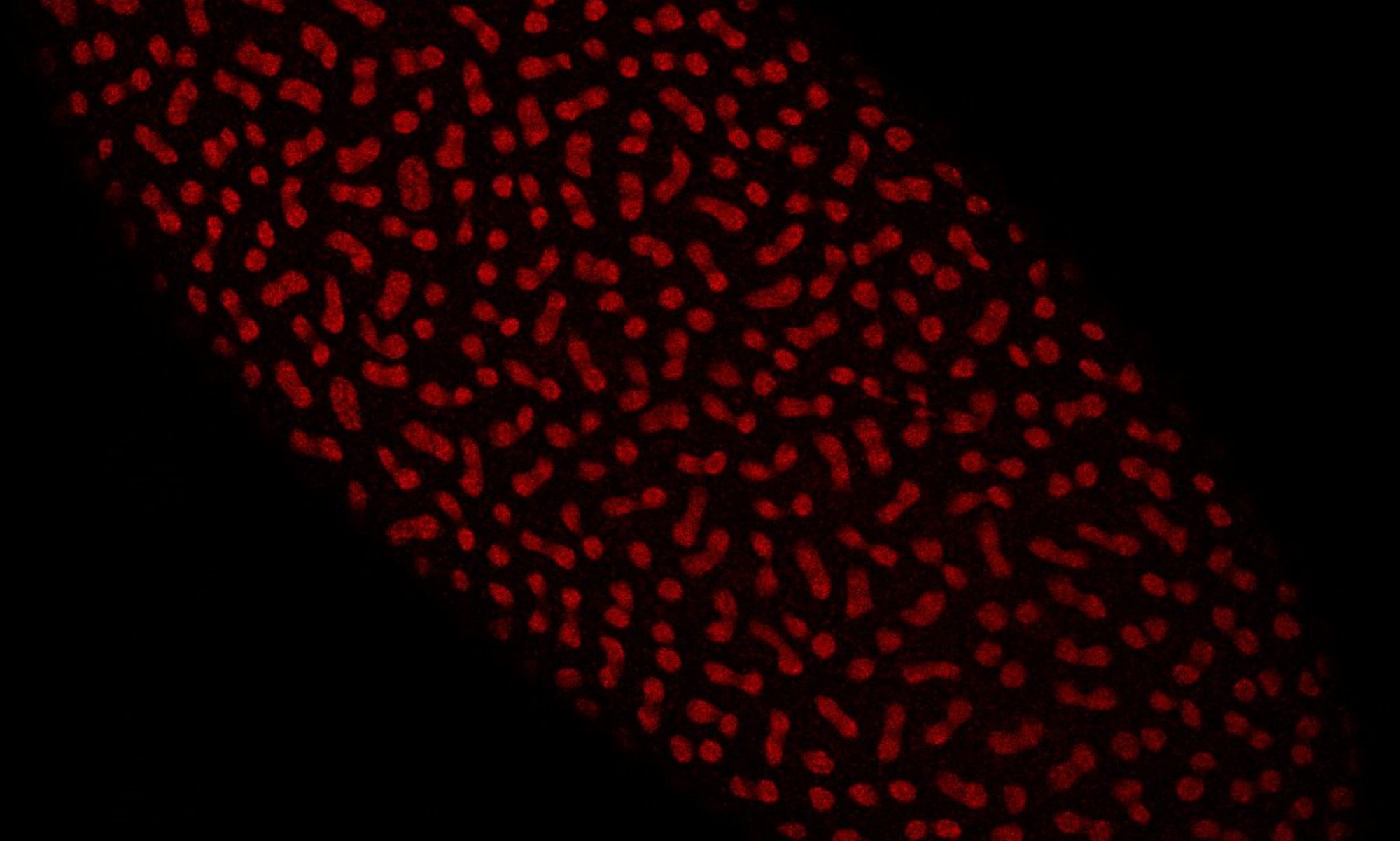
Lipid droplets play critical functions in the handling of specific proteins, affecting their maturation, storage, and turnover (Welte and Gould, 2017). One of the best characterized examples is the sequestration of specific histones by LDs in Drosophila ovaries and embryos (Li et al., 2012). Here, LDs perform two functions: they prevent degradation of histones synthesized during oogenesis (Stephenson et al., 2021), and for the variant histone H2Av, they also buffer its availability during early embryogenesis and thus reduce the rate of H2Av import into the nucleus (Johnson et al., 2018). We are now molecularly dissecting how H2Av exchanges between LDs, how and why the rate of exchange is developmentally regulated, and how lack of H2Av sequestration impacts nuclear functions and development.
H2Av-GFP localized to nuclei (larger circles) and lipid droplets (smaller circles) (Johnson et al., 2018).
Metabolism
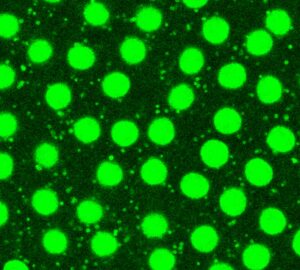
LDs are integral to lipid metabolic processes. This includes production of lipid signaling molecules, handling of fat-soluble vitamins, storage and release of fat , providing substrates for membrane synthesis, and numerous links to mitochondrial biology. LD metabolic biology is linked to obesity, diabetes, and fatty liver diseases, with emerging roles in dementia and other neurodegenerative diseases. We have found that during oogenesis LDs control the production of prostaglandins, lipid signaling molecules critical for oocyte maturation (Giedt et al., 2021). We are now characterizing how ovaries traffic fats to and from LDs and how faulty allocation of LDs between embryonic tissues disrupts metabolism and development.
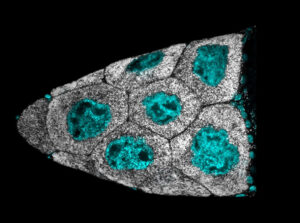
LDs (white) localized around nurse cell nuclei (cyan) in stage 10B Drosophila ovaries (Giedt et al., 2021).
Motility
Lipid droplets are motile in many cells (Kilwein & Welte 2019), with motion particularly vigorous in early Drosophila embryos. LDs move bidirectionally along microtubules, powered by the motors kinesin-1 and cytoplasmic dynein (Shubeita et al., 2008, Gross et al., 2000). Because many other intracellular cargoes move bidirectionally employing these same two motors, LDs are a great model for uncovering general transport mechanisms. For example, we discovered the motor regulator Klar because of its role in LD motility (Welte 1998) and could later show that it also controls the movement and positioning of nuclei and mRNPs (Elhanany-Tamir et al., 2012 and Gaspar et al., 2014). Recently, we found that for LDs to move correctly, they need to be kept apart from other nutrient-storing bodies (Kilwein et al., 2023).
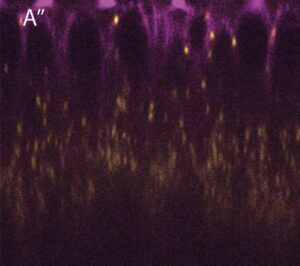
Lipid droplets (yellow) travel along microtubules (magenta) in Drosophila embryos (Kilwein and Welte, 2021).